AMINES
1. Mark the correct statement
(a) Methylamine is slightly acidic
(b) Methylamine is less basic than ammonia
(c) Methylamine is a stronger base than ammonia
(d) Methylamine forms salts with alkalies.
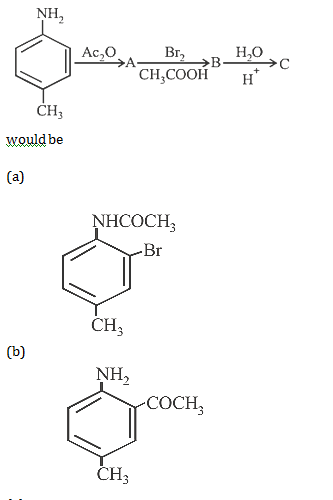
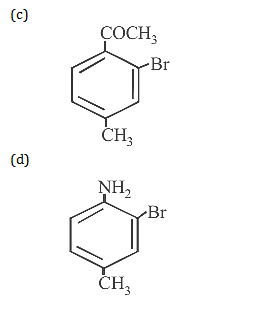
2. Indicate which nitrogen compound amongst the following would undergo Hofmann reaction (i.e.., reaction with Br2 and strong KOH) to furnish the primary amine (R– NH2)
(a) RCONHCH3
(b) RCOONH4
(c) RCONH2
(d) RCONHOH.
3. For carbylamine reaction, we need hot alcoholic KOH and
(a) Any primary amine and chloroform
(b) Chloroform and silver powder
(c) A primary amine and an alkyl halide
(d) A monoalkylamine and trichloromethane.
4. What is the decreasing order of basicity of primary, secondary and tertiary methylamines and NH3?
(a) NH3 > C2H5NH2 > (C2H5)2NH > (C2H5)3N
(b) (C2H5)3N > (C2H5)2NH > C2H5NH2 > NH3
(c) (C2H5)2NH > C2H5NH2 > (C2H5)3N > NH3
(d) (C2H5)2NH > (C2H5)3N > C2H5NH2 > NH3
5. Acetamide and ethylamine can be distinguished by reacting with
(a) Aqueous HCl and heat
(b) Aqueous NaOH and heat
(c) Acidified KMnO4
(d) Bromine water.
6. Which is formed, when acetonitrile is hydrolysed partially with cold concentrated HCl?
(a) acetic acid
(b) acetamide
(c) methyl cyanide
(d) acetic anhydrides
7. When aniline reacts with oil of bitter almonds (C6H5CHO) condensation takes place and benzal derivative is formed. This is known as
(a) Million’s base
(b) Schiff’s reagent
(c) Schiff’s base
(d) Benedict’s reagent
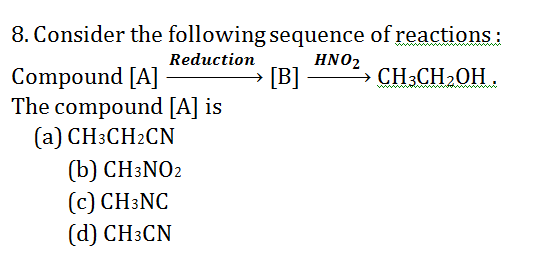
9. Aniline is an activated system for electrophilic substitution. The compound formed on heating aniline with acetic anhydride is
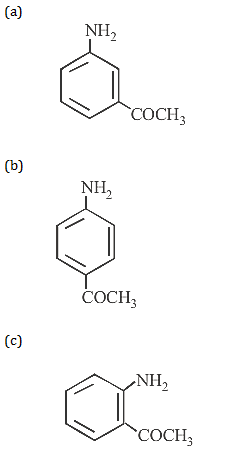
10. The compound obtained by heating a mixture of a primary amine and chloroform with ethanolic potassium hydroxide (KOH) is
(a) an alkyl cyanide
(b) a nitro compound
(c) an alkyl isocyanide
(d) an amide
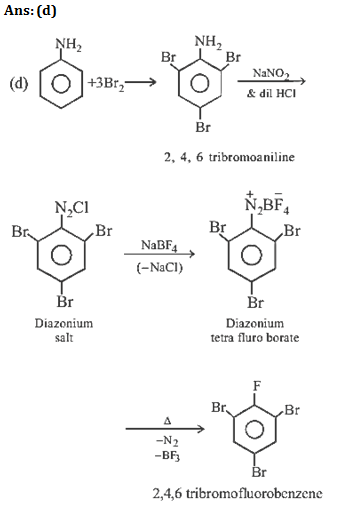
11. Aniline is reacted with bromine water and the resulting product is treated with an aqueous solution of sodium nitrite in presence of dilute
hydrochloric acid. The compound so formed is converted into a tetrafluoroborate which is subsequently heated dry. The final product is
(a) 1,3, 5-tribromobenzene
(b) p-bromofluorobenzene
(c) p-bromoaniline
(d) 2,4, 6-tribromofluorobenzene
12. An isocyanide is prepared by
(a) Friedel-Crafts reaction
(b) Perkin reaction
(c) Carbylamine reaction
(d) Wurtz reaction
13. Which of the following reagents will convert pmethylbenzenediazonium chloride into pcresol?
(a) Cu powder
(b) H2O
(c) H3 PO2
(d) C6H5OH
14. The consituent of the powerful explosive RDXis formed during the nitration of
(a) toluene
(b) phenol
(c) glycerol
(d) urotropine
15. Which of the following is most basic in nature?
(a) NH3
(b) CH3NH2
(c) (CH3) 2NH
(d) C6H5NHCH3
16. [A] [B] [C] N – Methylanil line , A is
(a) Formaldehyde
(b) Trichloromethane
(c) Nitrobenzene
(d) Toluene
17. Intermediates formed during reaction of with Br2 and KOH are
(a) RNHBr and RCONHBr
(b) RNHCOBr and RNCO
(c) RCONHBr and RNCO
(d) RCONBr2
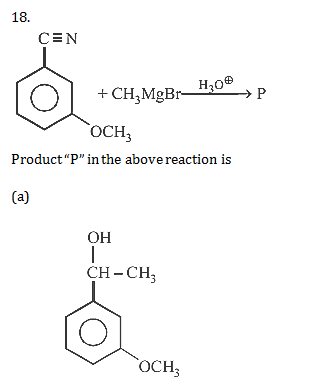
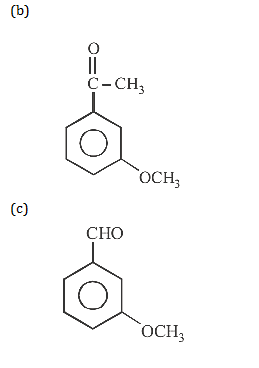
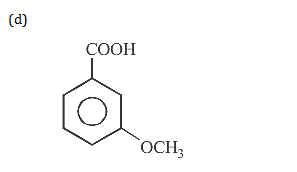
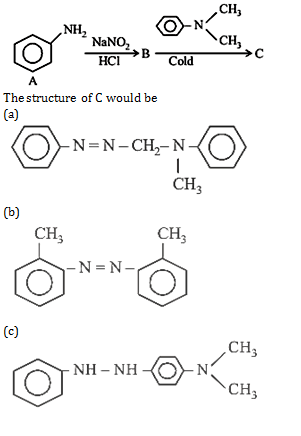
19. The final product C, obtained in this reaction
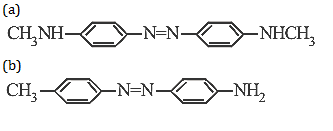
20. Aniline when diazotized in cold and when treated with dimethyl aniline gives a coloured product. Its structure would be
21. Aniline in a set of reactions yielded a product D.
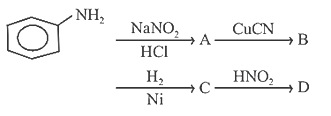
The structure of the product D would be
(a) C6H5NHOH
(b) C6H5NHCH2CH3
(c) C6H5CH2NH2
(d) C6H5CH2OH
22.The major organic product formed from the following reaction :
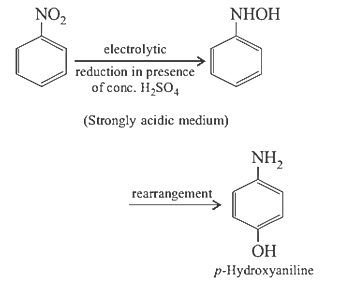
23. Electrolytic reduction of nitrobenzene in weakly acidic medium gives
(a) N-Phenylhydroxylamine
(b) Nitrosobenzene
(c) Aniline
(d) p-Hydroxyaniline
24. Which of the following is more basic than aniline?
(a) Triphenylamine
(b) p-Nitroaniline
(c) Benzylamine
(d) Diphenylamine
25. Which one of the following on reduction with lithium aluminium hydride yields a secondary amine?
(a) Methyl isocyanide
(b) Acetamide
(c) Methyl cyanide
(d) Nitroethane.
26. In a reaction of aniline a coloured product C was obtained.
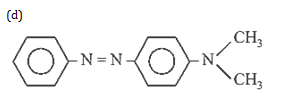
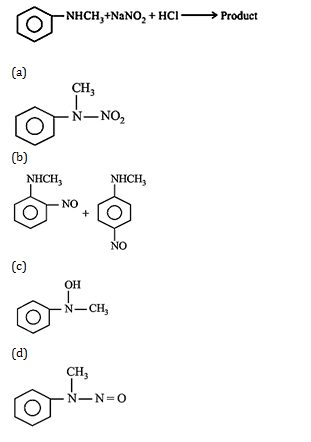
27. Predict the product :
28. Nitrobenzene can be prepared from benzene by using a mixture of conc. HNO3 and conc. H2 SO4 in the mixture, nitric acid acts as a/an:
(a) acid
(b) base
(c) catalyst
(d) reducing agent
29. Aniline in a set of the following reactions yielded a coloured product “Y”
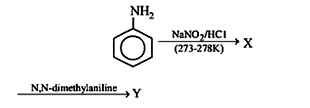
The structure of “Y” would be :
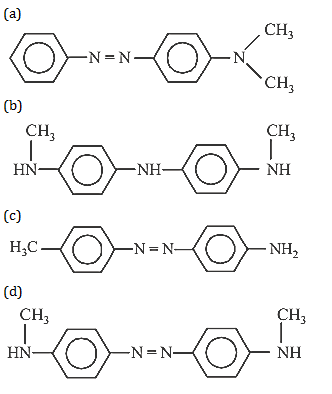
30. Acetamide is treated with the following reagents separately. Which one of these would yield methylamine?
(a) NaOH– Br2
(b) Sodalime
(c) Hot conc. H2SO4
(d) PCl5
31. Which of the following statements about primary amines is ‘False’?
(a) Alkyl amines are stronger bases than aryl amines
(b) Alkyl amines react with nitrous acid to produce alcohols
(c) Aryl amines react with nitrous acid to produce phenols
(d) Alkyl amines are stronger bases than ammonia
32. What is the product obtained in the following reaction :
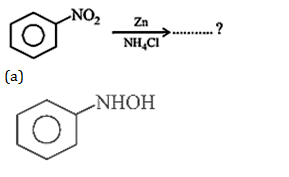
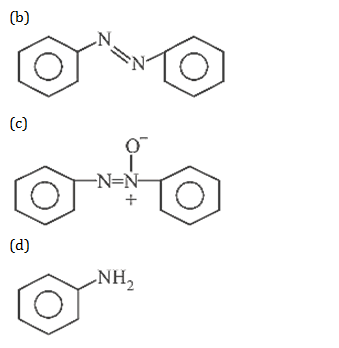
33. Which of the following compounds is most basic?
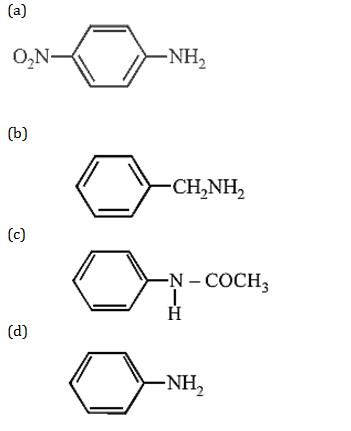
34. An organic compound (C3H9N) , when treated with nitrous acid, gave an alcohol and N2 gas was evolved. on warming with CHCl3 and caustic potash gave which on reduction gave isopropyl methylamine. Predict the structure of .
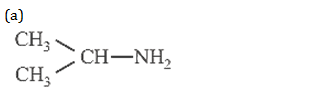
(b) CH3CH2 – NH – CH3
(d) CH3CH2CH2 – NH2
35. In the reaction
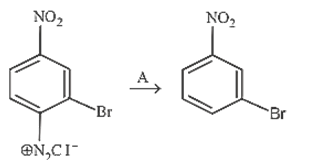
A is
(a) Cu2Cl2
(b) H3 PO2 and H2O
(c) H+ /H2O
(d) HgSO4 /H2 SO4
36. On hydrolysis of a “compound”, two compounds are obtained. One of which on treatment with sodium nitrite and hydrochloric acid gives a product which does not respond to iodoform test. The second one reduces Tollen’s reagent and Fehling’s solution. The “compound” is
(a) CH3 CH2 CH2CON(CH3) 2
(b) CH3 CH2 CH2NC
(c) CH3 CH2 CH2CN
(d) CH3 CH2 CH2ON = O
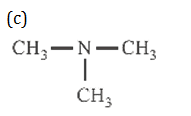
37.Some reactions of amines are given. Which one is not correct?
(a) (CH3)2NH +NaNO2 +HCl ® (CH3)2 N – N = 0
(c) CH3CH2NH2 +HNO2 ® CH3CH2OH +N2
(d) CH3NH2 +C6H5SO2Cl ® CH3NHSO2C6H5
38. When primary amine is heated with CS2 in presence of excess of HgCl2 , it gives isocyanate The reaction is called :
(a) Hoffmann’s bromamide reaction
(b) Carbylamine reaction
(c) Perkin’s reaction
(d) Hoffmann’s mustard oil reaction
39. Which one of the following nitro-compounds does not react with nitrous acid?
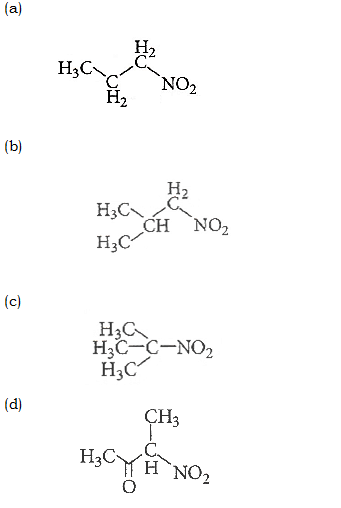
40. The correct statement regarding the basicity of arylamines is
(a) arylamines are generally more basic than alkylamines because of aryl group
(b) arylamines are generally more basic than alkylamines because the nitrogen lone-pair electrons are not delocalised by interaction with the aromatic ring π-electron system
(c) arylamines are generally more basic than alkylamines, because the nitrogen atom in arylamines is sp-hybridised
(d) arylamines are generally less basic than alkrylamines because the nitrogen lone-pair electrons are delocalised by interaction with the aromatic ring π-electron system
41. The method by which aniline cannot be prepared is ……………
(a) degradation of benzamide with Br2 / NaOH
(b) reduction of nitrobenzene by Sn / HCI
(c) potassium salt of phthalimide treated with chlorobenzene followed by hydrolysis with aqueous NaOH solution
(d) Hydrolysis of phenylcyanide with acidic solution
42. Nitrobenzene gives N phenylhydroxylamine by
(a) Sn/HCl
(b) Zn/NH4Cl
(c) H2/Pd-C
(d) Zn/NaOH
43. The electrolytic reduction of nitrobenzene in strongly acidic medium produces
(a) azoxybenzene
(b) azobenzene
(c) aniline
(d) p-aminophenol
44. Which of the following will be most stable diazonium salt R X– ?
(a) CH3 X-
(b) C6H5 X-
(c) C6H5CH2 X-
(d) CH3CH2N2+X-
45. On hydrolysis of a “compound”, two compounds are obtained. One of which on treatment with sodium nitrite and hydrochloric acid gives a product which does not respond to iodoform test. The second one reduces Tollens reagent and Fehling’s solution. The “compound” is
(a) CH3CH2CH2NC
(b) CH3CH2CH2CN
(c) CH3CH2CH2CON(CH3)2
(d) CH3CH2CH2ON=O
46. Which is the most suitable reagent among the following to distinguish compound (3) from rest of the compounds?
(1) CH3 – C ≡ C – CH3
(2) CH3 – CH2 – CH2 – CH3
(3) CH3 – CH2C ≡ CH
(4) CH3 – CH = CH2
(a) Bromine in carbon tetrachloride
(b) Bromine in acetic acid
(c) Alk. KMnO4
(d) Ammoniacal silver nitrate
47. Nitrobenzene can be prepared from benzene by using a mixture of conc. HNO3 and conc. H2SO4. ln the mixture, nitric acid acts as a/an
(a) acid
(b) reducing agent
(c) base
(d) catalyst
48. Aniline in a set of reactions yielded a product D.
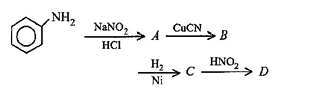
The structure of the product D would be
(a) C6H5NHOH
(b) C6H5CH2OH
(c) C6H5NHCH2CH3
(d) C6H5CH2NH2
49. Intermediates formed during reaction of RCONH2 with Br2 and KOH are
(a) RCONBr2
(b) RCONHBr and RNCO
(c) RNHCOBr and RNCO
(d) RNH – Br and RCONHBr
50. Phenyl isocyanides are prepared by which of the following reaction?
(a) Reimer-Tiemann reaction
(b) Wurtz reaction
(c) Carbylamine reaction
(d) Rosenmund’s reaction
51. Diethyl oxalate is used for distinguishing primary, secondary and tertiary
(a) alkyl halides
(b) hydrogens in hydrocarbons
(c) alcohols
(d) amines
52. Which product is formed, when acetonitrile is hydrolysed partially with cold concentrated HCl?
(a) Methyl cyanide
(b) Acetamide
(c) Acetic anhydride
(d) Acetic acid
53. Which one of the following order is wrong, with respect to the property indicated?
(a) Formic acid > acetic acid > propanoic acid (acid strength)
(b) Fluoroacetic acid > chloroacetic acid > bromoacetic acid (acid strength)
(c) Benzoic acid > phenol > cyclohexanol (acid strength)
(d) Aniline > cyclohexylamine > benzamide (basic strength)
54. Indicate which nitrogen compound amongst the following would undergo Hofmann reaction (i.e., reaction with Br2 and strong KOH) to furnish the primary amine (R-NH2)?
(a) RCONHCH3
(b) R-CO-NHOH
(c) RCOONH4
(d) RCONH2
55. Pyridine is less basic than triethylamine because
(a) pyridine is a cyclic system
(b) in pyridine, lone pair of electrons on nitrogen is delocalized
(c) pyridine has aromatic character
(d) nitrogen in pyridine is sp2-hybridised
56. Nitrobenzene can be prepared fiom benzene by using a mixture of conc. HNO3 and conc. H2SO4. ln the mixture, nitric acid acts as a/an
(a) acid
(b) base
(c) catalyst
(d) reducing agent
57. For carbylamines reaction , we need hot alcoholic KOH and
(a) any primary amine and chloroform
(b) chloroform and silver powder
(c) a primary amine and an alkyl halide
(d) a moanalkylimine and trichloromethane.
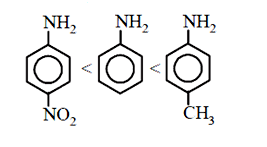
58. The correct increasing order of basic strength for the following compounds is :
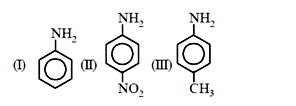
(a) III < I < II
(b) III < II < I
(c) II < I < III
(d) II < III < I
59. Which of the following reactions is appropriate for converting acetamide to methanamine ?
(a) Hoffmarnn hypobromamide reaction
(b) Stephens reaction
(c) Gabriels phthalimide synthesis
(d) Carbylamine reaction
60. Nitration of aniline in strong acidic medium also gives m-nitroaniline because
(a) Inspite of substituents nitro group always goes to only m-position.
(b) In electrophilic substitution reactions amino group is meta directive.
(c) In acidic (strong) medium aniline is present as anilinium ion.
(d) In absence of substituents nitro group always goes to m-position
61. The correct order of the basic strength of methyl substituted amines in aqueous solution is :
(a) (CH3)2 NH > CH3NH2 > (CH3)3N
(b) (CH3)3N > CH3NH2 > (CH3)2 NH
(c) (CH3)3N > (CH3)2 NH > CH3 NH2
(d) CH3NH2 > (CH3)2NH > (CH3)3N
62. Which of the following amine will give the carbylamine test?
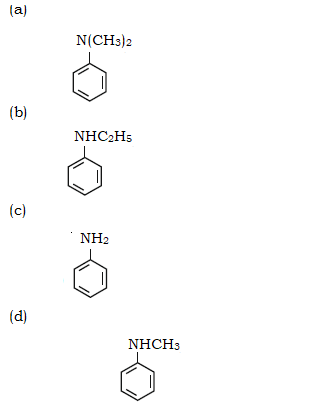
63. Identify the compound that will react with Hinsberg’s reagent to give a solid which dissolves in alkali.
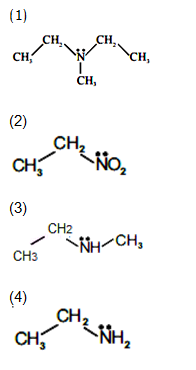
64. The RBC deficiency is deficiency disease of :
(a) Vitamin B2
(b) Vitamin B12
(c) Vitamin B6
(d) Vitamin B1
1.Ans: (c) Methyl amine is a stronger base than ammonia due to +I effect. The alkyl groups which are electrons releasing groups increase in the electron density around the nitrogen thereby increasing the availability of the lone pair of electrons to proton or lewis acid and making the amine more basic
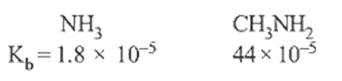
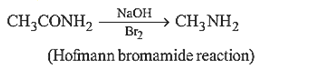
2.Ans: (c) Only 10 amides(i.e. RCONH2) in the present case undergo Hoffmann bromamide reaction.
RCONH2 +Br2 +4KOH R – NH2 +2KBr + K2CO3 +2H2O
(Hoffman’s bromamide reaction)
3.Ans: (a) Any primary amine means both aliphatic as well as aromatic but monoalkylamines means only 10 aliphatic amines. Therefore, option (a) is correct while (d) is wrong.
4.Ans: (d) All aliphatic amines are stronger bases than NH3 and among different ethylamines order of basicity is 20 > 30 >10 . Thus , the correct order is (d) i.e.
(C2H5)2 NH > (2H5)3 > C2H5NH2 >NH3.
This anomalous behaviour of teritiary ethyl amine is due to steric factors. i.e., crowding of alkyl groups cover nitrogen atom from all sides and thus make the approach and bonding by a lewis acid relatively difficult which results the maximum steric strain in tertiary amines. The electrons are there but the path is blocked resulting the reduction in its basicity.
5.Ans: (b) Acetamide and ethylamine can be distinguished by heating with NaOH solution.
Acetamide evolves NH3 but ethylamine does not.
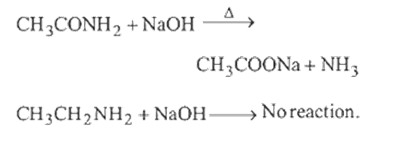
6.Ans: (b) Methyl cyanide on treatment with conc. HCl give acetamide
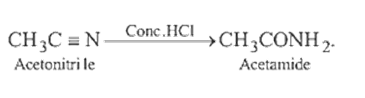
7.Ans: (c) Benzaldehyde reacts with primary aromatic amine to form schiff’s base
This is known as Schiff’s base reaction.
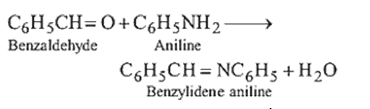
8.Ans: (d)
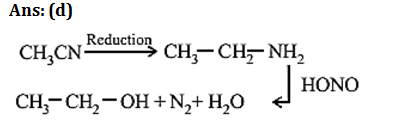
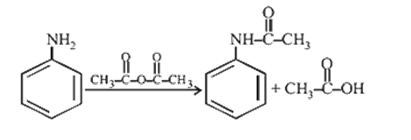
9.Ans: (d) Aniline when treated with acetic anhydride forms acetanilide (nuclephilic substitution)
10.Ans: (c) CH3NH2 +CHCl3 -> CH3CH2NC +3KCl +3H2O In this reaction, bad smelling compound ethyl isocyanide (CH3CH2NC) is produced. This equation is known as carbyl amine reaction.
11.Ans: (d)
12.Ans: (c)
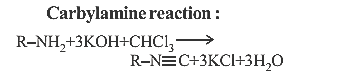
13.
14.Ans: (d) RDX is prepared by treating urotropine with fuming nitric acid. When the inner bridge system isdestroyed by oxidationadn the peripheral nitrogen atom are nitrated, it forms cyclonitrite( or RDX).
15.Ans: (c)
(CH3)2 NH is most basic because two electron releasing groups are present on Nitrogen. Also aromatic amines are less basic then aliphatic amines. The basic character of amines follow the order
R2NH>RNH2> C6H5NHCH3 >NH3
16.
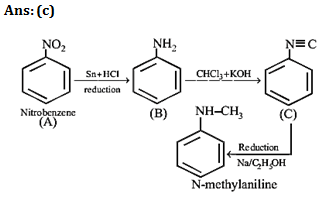
17.
18.Ans: (b)
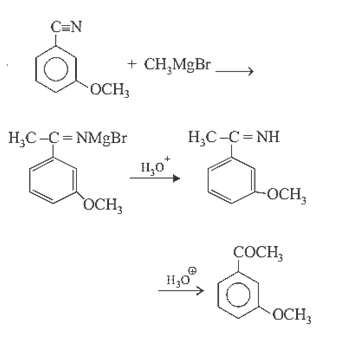
19.Ans: (d)
20.Ans: (c)
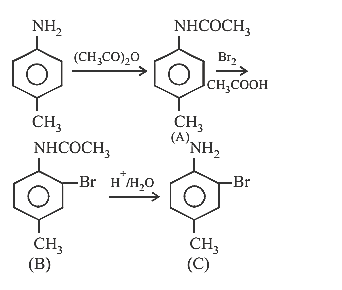
21.Ans: (d)
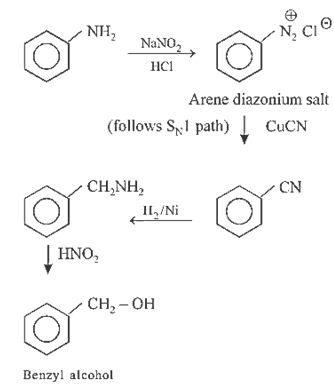
22.Ans: (b)
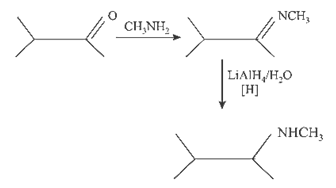
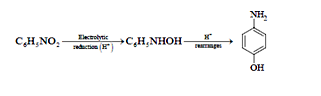
23.Ans: (c) Electrolytic reduction of nitroalkane in weakly acidic medium give aniline

Whereas in strongly acidic medium it gives p –hydroxyaniline.
24.Ans: (c) Benylamine is more basic than aniline. The reason is that in aniline, the lone pair of nitrogen is conjutgated with benzene ring so it is not available readily for others. On the other hand in Benzylamine, nitrogen is not directly attached with ring so lone pairs are not conjugated with ring.
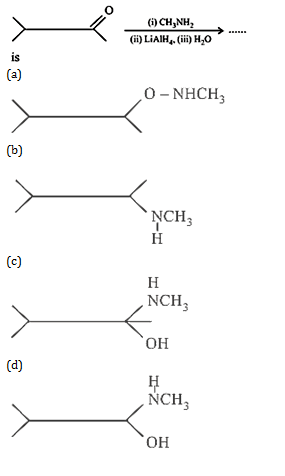
25.Ans: (a) Reduction of alkyl isocynides in presence of LiAlH4 yields secondary amines containing methyl as one of the alkyl group. Whereas alkyl cyanides give 10 amine on reduction.
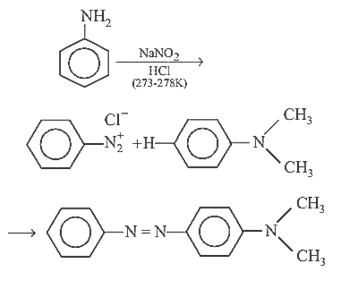
26.Ans: (d) When aniline is reacted with NaNO2 and HCl it gives benzene diazonium chloride This benzene diazonium chloride on coupling with dimethylaniline gives coloured compound I.e p-(N,N-dimethyl) amino azobenzene.
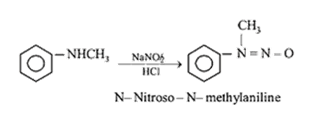
27.Ans: (d)Secondary amine with (NaNO2 +HCl) gies a nitroso product
28.Ans: (b) HONO2 + H2SO4 ® +H2O + Nitric acid acts as a base by accepting a proton.
29.Ans: (a)
30.Ans: (a)
31.Ans: (c)Aryl amines do not produce phenol on treatement with nitrous acid.
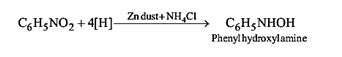
32.Ans: (a)When nitro compound is reduced with a neutral reducing agent (Zn dust +NH4Cl) the corresponding hydroxyl amine is formed.
33.Ans: (b) Compound is most basic due to localized lone pair of electron on nitrogen atom while other compounds have delocalized lone air of electron.
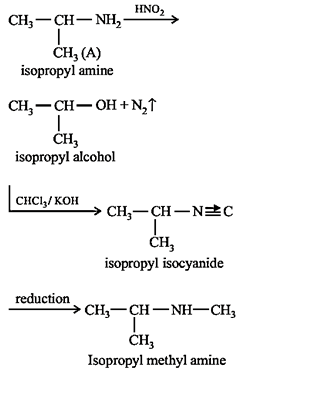
34.Ans: (a)
35.Ans: (b)
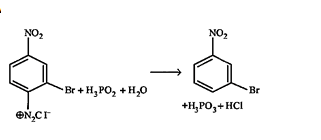
36.Ans: (b) Hydrolysis gives CH3CH2CH2NH2+HCOOH. On treatment with NaNO2 and HCl former gives CH3CH2CH2OHG which does not give iodoform test. HCOOH gives tollen’s reagent test.
37.Ans: (b) Secondary amine react with nitrous acid to give N – nitrosoamines.
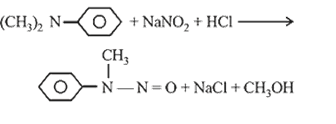
38.(c) Perkin’s reaction
39.(c) Tertiary nitroalkanes do not react with nitrous acid as they do not contain α-hydrogen atom.
40.(d) arylamines are generally less basic than alkrylamines because the nitrogen lone-pair electrons are delocalised by interaction with the aromatic ring π-electron system
41.(c) Aniline cannot be prepared by this method because aryl halides do not undergo nucleophilic substitution reaction with potassium phthalimide under mild conditions.
42.(b) Reduction of nitrobenzene in neutral medium [zinc dust and ammonium chloride or acetic acid sodium acetate] converts nitrobenzene into N-phenylhylamine
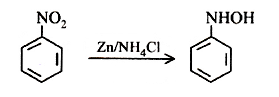
43 (d)
44. (b) Diazonium salt containing aryl group directly linked to the nitrogen atom is most stable due to resonance stabilisation between the benzene nucleus and N-atom. Diazonium ion act as a electrophile.[Resonance structure of benzene diazonium ion]
45.(a) CH3CH2CH2NC
46.(d) All the three reagents except ammoniacal AgNO3 reacts with 1, 2 and 4 compounds. The compound 3 possessing the terminal alkyne only reacts with ammoniacal AgNO3 and thus can be distinguished from 1, 2 and 4 compounds.
47.(c)
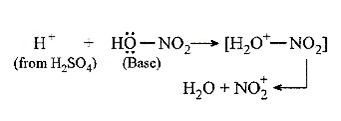
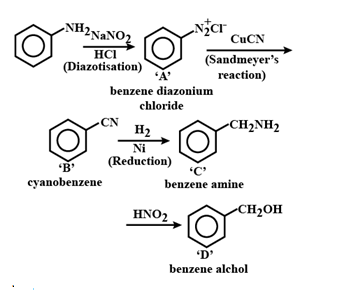
48. (b)
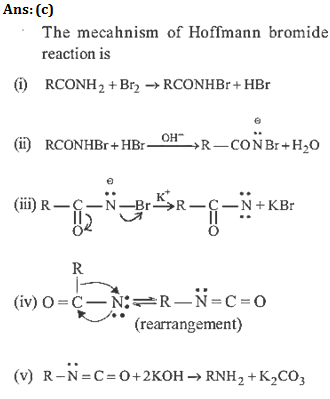
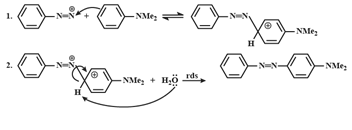
49.(b) The reaction RCONH2 +Br2 + KOH → RNH2 is known as Hoffmann – bromamide reaction. The mechanism of the reaction is
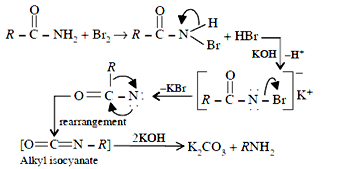
This reaction is used in the descent of series. i.e. for preparing a lower homologue from a higher one.
50 (b) C6H5 – NH2 + CHCI3 + 3KOH → C6H5– NC + 3KCl + 3H2O The above reaction is called as carbylamine reaction, which is a specific reaction of 1° amine. In this reaction, aniline is treated with an alkaline solution of chloroform to give phenyl isocyanide.
51 (d) amines
52 (b)
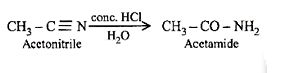
53(d) Cyclohexylamines are more basic than aniline, the later shows resonance.
54(d) The amide (-CONH2) group is converted into a primary amino group (-NH2) by Hofmann’s bromamide reaction.
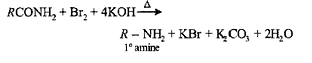
55. (b) Basicity of amines is due to availability of an unshared pair (lone pair) of electrons on nitrogen. This lone pair of electrons is available for the formation of a new bond with a proton or Lewis acid. Pyridine is less basic than triethylamine because lone pair of nitrogen in pyridine is delocalised.
56(b)
57(a) In carbylamine reaction, primary amines on heating with chloroform in presence of alcoholic KOH form isocyanides (or carbylamines). It is used to distinguish 1° amines from 2° and 3° amines.
58 (c) Order of basic strength
59 (a)
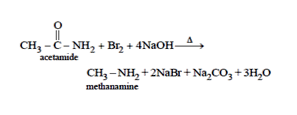
60 (c) In acidic medium, aniline is proronated to form anilium ion which is meta directing, Hence besides para (51%)and ortho (2%) meta product (47%)is also formed in significant yield.
61 (a) (CH3)2 NH > CH3NH2 > (CH3)3N
62 (c)
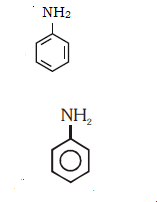
Since is primary amine it gives carbylamines test (isocyanide test)
63 (d)1° amines react with Hingsberg’s reagent to give a solid, which dissolve in alkali.
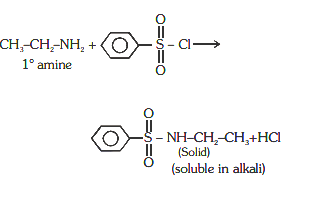
64 (b)Deficiency of vitamin B12 causes Megaloblastic Anaemia/Pernicious anaemia.












