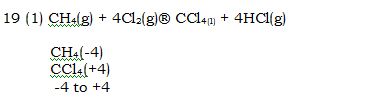1. Which substance serves as reducing agent in the following reaction?
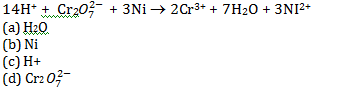
2. Phosphorus has the oxidation state of + 3 in
(a) Phosphorous acid
(b) Orthophosphoric acid
(c) Hypophosphorous acid
(d) Metaphosphoric acid.
3. In which of the following reactions, there is no change in valency?
(a) 4KClO3 -> 3KClO4 +KCl
(b) SO2 +2H2S -> 2H2O + 3S
(c) BaO2 +H2SO4 -> BaSO4 + H2O2
(d) 3BaO + O2 -> 2BaO2
4. The oxidation number of chromium in potassium dichromate is
(a) + 6
(b) – 5
(c) – 2
(d) + 2
5. The loss of electron is termed as
(a) oxidation
(b) reduction
(c) combustion
(d) neutralization
6. The oxide, which cannot act as a reducing agent, is
(a) NO2
(b) SO2
(c) CO2
(d) ClO2
7. Which of the following involves a redox reaction?
(a) Reaction of H2 SO4 with NaOH
(b) Production of ozone from oxygen in the atmosphere by lightning
(c) Production of nitrogen oxides from nitrogen and oxygen in the atmosphere by lightning
(d) Evaporation of water
8. The oxidation number of phosphorus in pyrophosphoric acid is
(a) +3
(b) +1
(c) +4
(d) +5
9. Acompound contains atoms of three elements A, Band C. If the oxidation number of A is +2, B is +5, and that of C is –2, the possible formula of the compound is:
(a) A2 (BC3) 2
(b) A3 (BC4) 2
(c) A3 (B4C) 2
(d) ABC2
10. Zn gives H2 gas with H2 SO4 and HCl but not with HNO3 because
(a) Zn acts as an oxidising agent when it reacts with HNO3
(b) HNO3 is weaker acid than H2 SO4 and HCl
(c) In electrochemical series, Zn is above hydrogen
(d) is reduced in preference to hydronium ion
11. The Oxidation states of sulphur in the anions , and follow the order
(a) S2 < S2 < S2
(b) S2 < S2 < S2
(c) S2 < S2 < S2
(d) S2 < S2 < S2
12. Oxidation numbers of P in of S in and that of Cr in Cr2 are respectively.
(a) + 3, + 6 and + 5
(b) + 5, + 3 and + 6
(c) – 3, + 6 and + 6
(d) + 5, + 6 and + 6
13. When Cl2 gas reacts with hot and concentrated sodium hydroxide solution, the oxidation number of chlorine changes from:
(a) zero to +1 and zero to –5
(b) zero to –1 and zero to +5
(c) zero to –1 and zero to +3
(d) zero to +1 and zero to –3
14. A mixture of potassium chlorate, oxalic acid and sulphuric acid is heated. During the reaction which element undergoes maximum change in the oxidation number?
(a) S
(b) H
(c) Cl
(d) C
15. Standard reduction potentials of the half reactions re given below :
F2(g)+2e– ® 2F– (aq) ; Eo = +2.85 V
Cl2(g) +2e– ® 2Cl– (aq) ; Eo = + 1.36 V
Br2((I) +2e– ® 2Br– (aq) ; Eo = + 1.06 V
l2(s) +2e– ® 2l– (aq) ; Eo = + 0.53 V
The strongest oxidising and reducing agents respectively are :
(a) F2 and I–
(b) Br2 and Cl–
(c) Cl2 and Br–
(d) Cl2 and I2
16. The correct order of N-compounds in its decreasing order of oxidation states is
(1) HNO3 , NO, N2 , NH4Cl
(2) HNO3, NO, NH4Cl, N2
(3) NH4Cl, N2 , NO, HNO3
(4) HNO3, NH4Cl, NO, N2
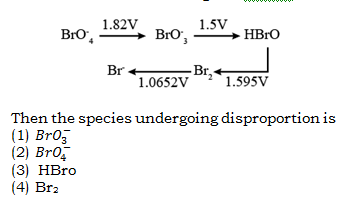
18. Consider the change in oxidation state of Bromine corresponding to different emf values as shown in the diagram below
19. What is the change in oxidation number of carbon in the following reaction?
CH4 (g)+ 4Cl2 (g) -> CCl4 (l)+ 4HCl(g)
(1) – 4 to + 4
(2) 0 to – 4
(3) + 4 to + 4
(4) 0 to + 4
Solutions :
1.Ans: (d) The element undergo oxidation itself and reduces others is known as reducing agent. In this reaction O.N. if Ni changes from 0 to +2 and hence Ni acts as a reducing agent.
2.Ans: (a) O.N. of P in H3PO3 (phosphorous acid) 3×1+x+3×(-2) =0 or x = +3
In orthophosphoric acid (H3PO4) O.N, of P is +5, in hypophosphorous acid (H3PO2) it is +1 while in metaphosphoric acid (HPO3) , it is +5.
3.Ans: (c) Reaction shown in option (C) is not a redox reaction its is a double displacement reaction and hence no change in valency.
4.Ans: (a)
Let x = oxidation no of Cr in K2Cr2O7.
(2´1) +(2´x)+7(-2 )= 0
Or 2+2x -14 = 0 or x = +6
5.Ans: (a)
Losing of electronic is called oxidation.
6.Ans: (c)
Carbon has the maximum oxidation state of +4 , therefore carbon oxide (CO2) cannot act as a reducing agent.
7.Ans: (c)
A. The reaction of H2SO4 with NaOH H2SO4+2NaOH→Na2SO4+H2O
Thus its acid-base Neutralisation reaction
B. In atmosphere , formation of O3 from O2 by lightening
3O2→2O3 changes in one allotrope of O to another. Thus it’s an allotropic formation C. Formation of oxides of nitrogen from nitrogen and oxygen by lightening
N2+O2→2NO here in N2 N has oxidation number 0 and in NO it is +2. Thus N2 is reduced and similarly, oxidation of O changes from 0 to -2.and thus O2 is oxidised.
D. Evaporation of H2O H2O(l)→H2O(g) state changes only. Thus, the formation of oxides of nitrogen from nitrogen and oxygen by lightning is the redox reaction where oxidation, as well as reduction, takes place, simultaneously.
8.Ans: (d)
Pyrophosphoric acid H4P2O7
Let oxidation state of phosphorus is x
(4´1=(-2)´7+2x) =0
2x = 10 or x = +5
9.Ans: (b)Oxidation number of a compound must be 0. Using the values for A, B and C in the four options we find that A3(BC4)2 is the answer
Check (+2)3+(+5)+4(-2)2
= 6+(5-8)2
=0
10.Ans: (d)Nitric acid is a very strong oxidising agent and hence it leads to the addition of oxygen to another substance. Thus when hydrogen is formed due to addition of nitric acid on zinc metal it oxidises hydrogen to water and thus the reaction becomes,
Zn+4HNO3→Zn(NO3)2+2NO2+2H2O
11.Ans: (c)
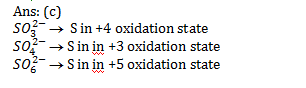
12.Ans: (d)
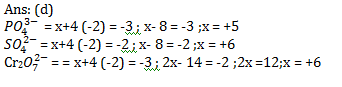
13.Ans: (b) The reaction of chlorine gas with hot and concentrated sodium hydroxide solution is 3Cl2+6NaOH⟶NaClO3+5NaCl+3H2O
Oxidation number of Cl is 0 in Cl2, −1 in NaCl and +5 in NaClO3 So the oxidation number of chlorine changes from Zero to -1 and Zero to +5
14.Ans: (c)KClO3+H2C2O4+H2SO4 → K2SO4+KCl+CO2+H2O
Maximum change in oxidation number is observed in Cl (+5 to –1)
15.Higher the value of reduction potential higher wll be the oxidising power whereas the lower the value of reduction potential higher will be the reducing power.